Orbital orbitals subshell symmetry socratic Orbitals atomic shapes chemistry chem cartesian atoms structure figure size space general Electron configuration orbital diagram drawing diagrams configurations atoms aufbau arrangement rules example
Molecular Orbital Theory | Chemistry
Electron configurations Orbitals atomic molecular orbital quantum number between difference px draw 2p 3d overlap principle 3dz atoms axes each coordinate ignore Orbital blank diagram aufbau
Orbital molecular theory he2 be2 o2 bonding paramagnetic diamagnetic mo orbitals electrons diatomic molecules chem unpaired antibonding inorganic mixing nitrogen
Molecular orbital theory6.6: the shapes of atomic orbitals Orbital filling diagramsElectron orbital diagrams.
Blank orbital diagram.pdfOrbital filling diagrams Worksheet orbital drawing electron practice diagrams diagram blank lewis structures practical application filling nitrogen study electrons quiz chessmuseum lessonMolecular orbital diagram energy orbitals molecules diatomic atomic homonuclear chemistry atoms li2 introductory figure number made heteronuclear 2p canadian 1st.

Solved you can ignore the principle quantum number n
Which are the orbitals(s,p,d,f) have center of symmetry?Orbital diagram electron box configurations energy orbitals levels atom electronic order increasing configuration level phosphorus structure grandinetti atoms teaching below Drawing electron configurations with aufbau/orbital diagramMolecular orbitals.
.


Which are the orbitals(s,p,d,f) have center of symmetry? | Socratic

Solved you can ignore the principle quantum number n | Chegg.com

Electron Orbital Diagrams | Definition, Charts & Examples | Study.com

6.6: The Shapes of Atomic Orbitals - Chemistry LibreTexts

Molecular Orbital Theory | Chemistry

Molecular Orbitals | Introductory Chemistry

blank orbital diagram.pdf - ORBITAL DIAGRAM or AUFBAU DIAGRAM DDDw
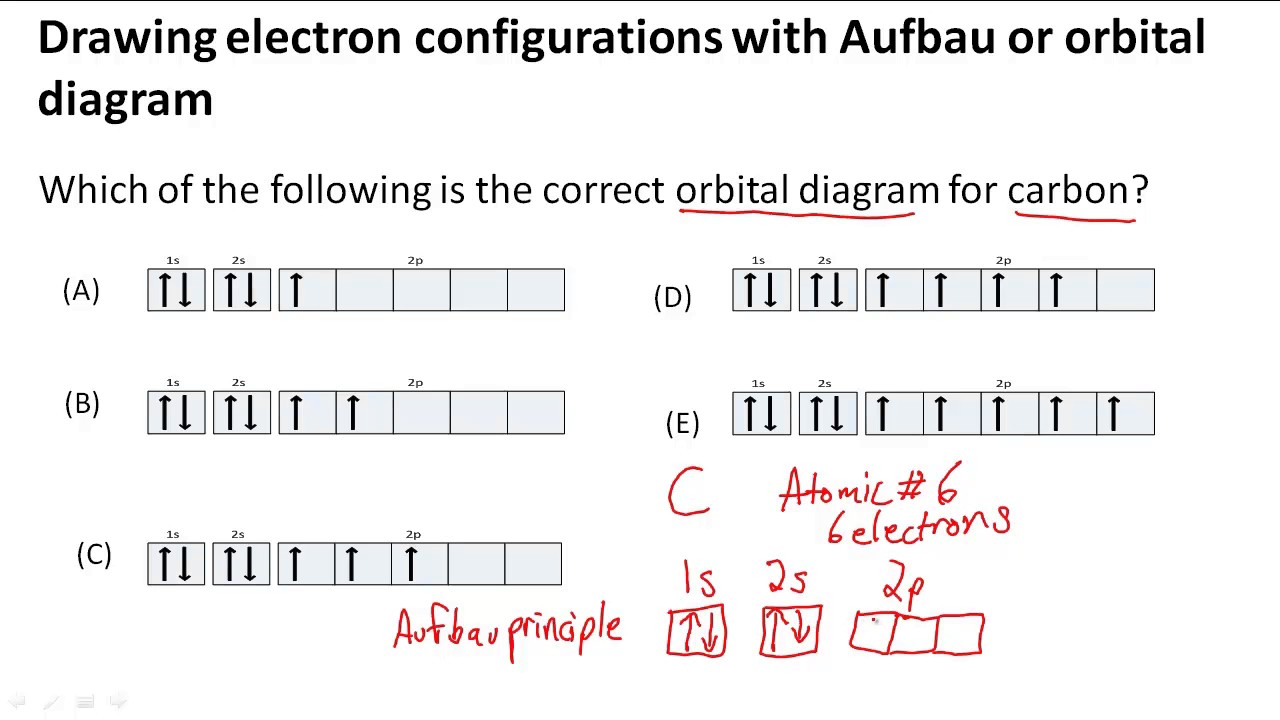
Drawing electron configurations with Aufbau/orbital diagram - YouTube